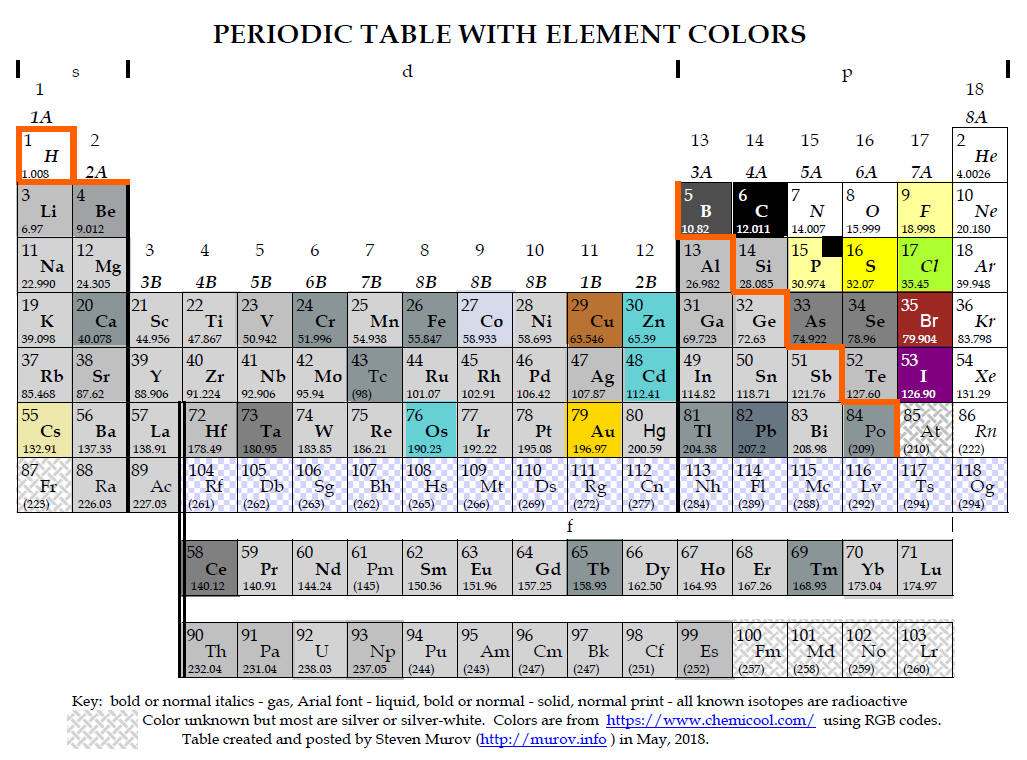
Kapische? Let’s go! Electromagnetic Radiation – Essential Knowledge 1.D.3 We will dive into an explanation about the quantum numbers and their physical interpretations, and we will finish up this article with an overview of how atomic structure influences periodicity and periodic trends in the periodic table. We’ll discuss Bohr’s model of the atomic structure of the hydrogen atom and the quantum mechanical model for atomic structure. We will then go on to discuss the particle-nature of matter and how the discoveries in the early 20 th century shaped our understanding of the atomic structure and spectrum of the hydrogen atom. 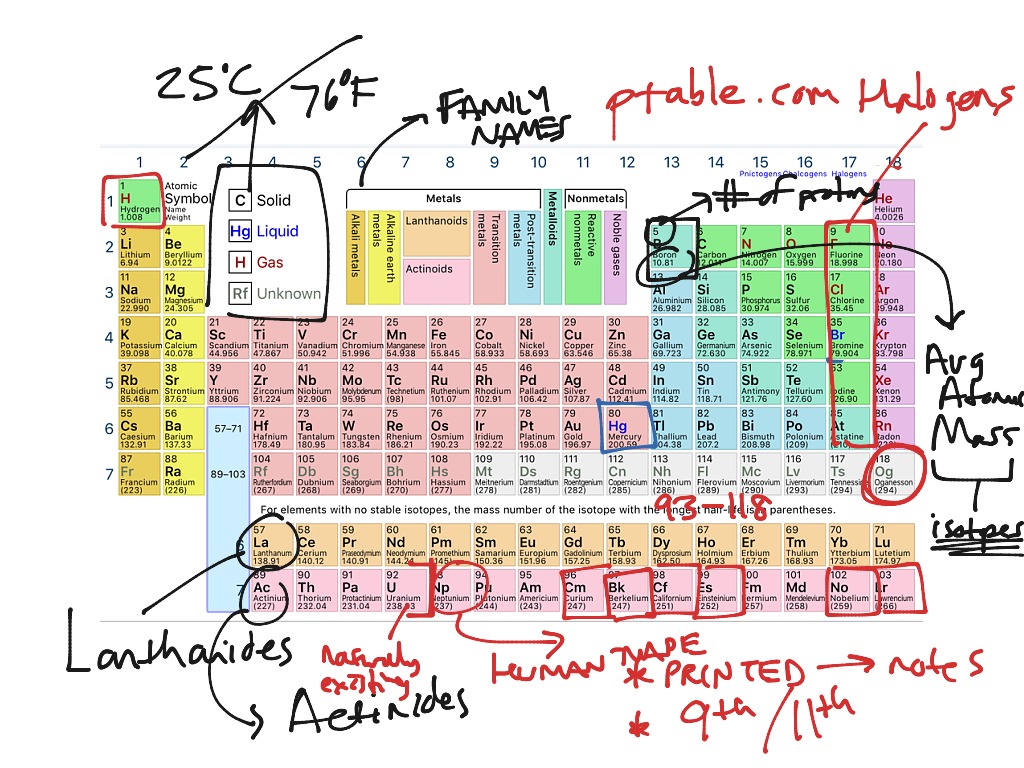
We will first start our discussion with a brief overview of the wave-like nature electromagnetic radiation. But what are atoms? What do they look like? And more importantly, how can you understand atomic structure for your AP® Chemistry Exam? In this Ultimate Guide, we’re going to give you a little bit of background on each topic to get you excited about it, and then we’re going to give you tips and advice on what you should memorize (or not), how to eliminate wrong answers quickly, and keywords that you should use in the long- and short-answer section of the exam. The chair you’re sitting on, the screen you’re looking at, and even the eyes you see with are made up of many different kinds of atoms. Having grown up in the 21 st century, you probably know that everything around you is fundamentally made of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
